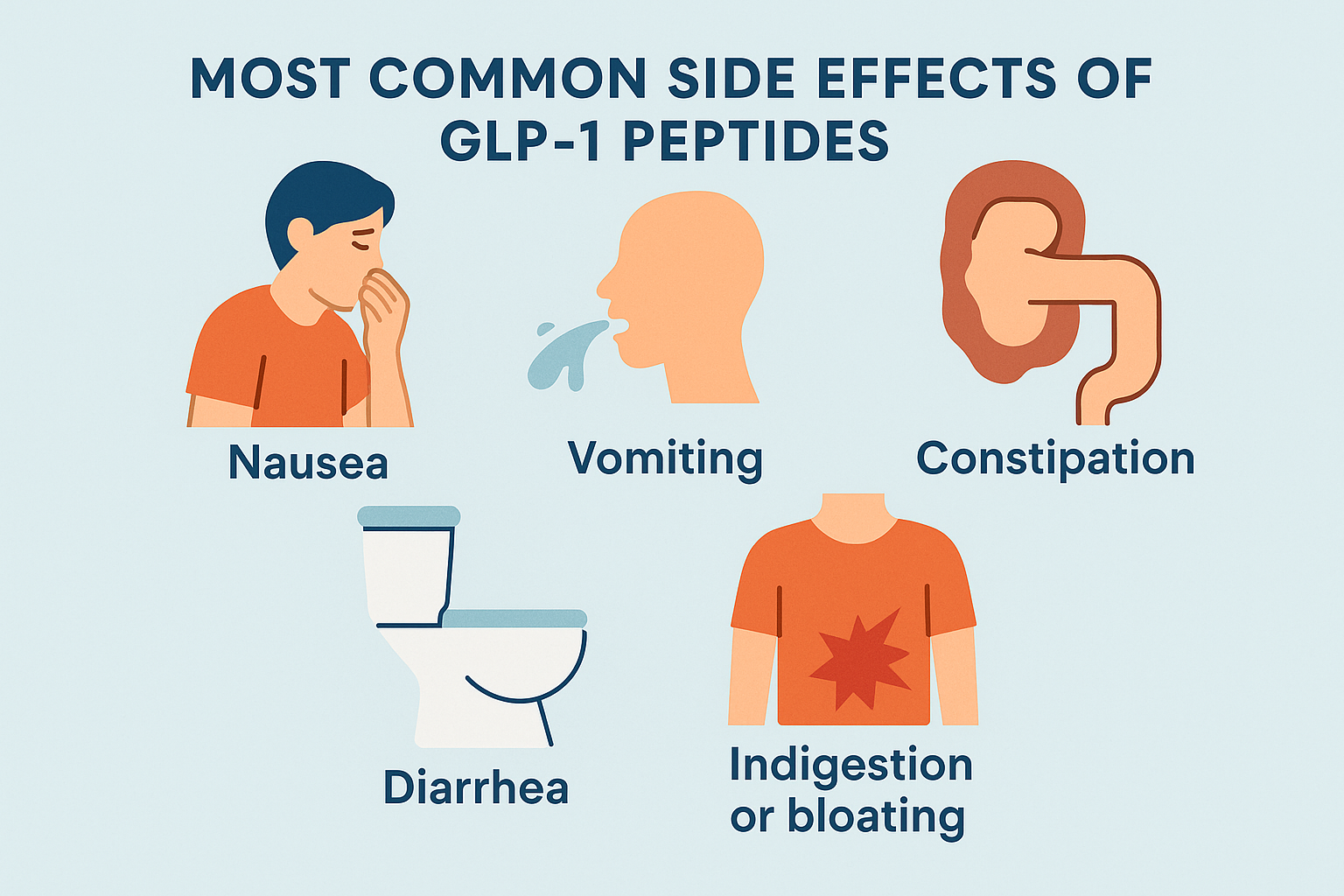
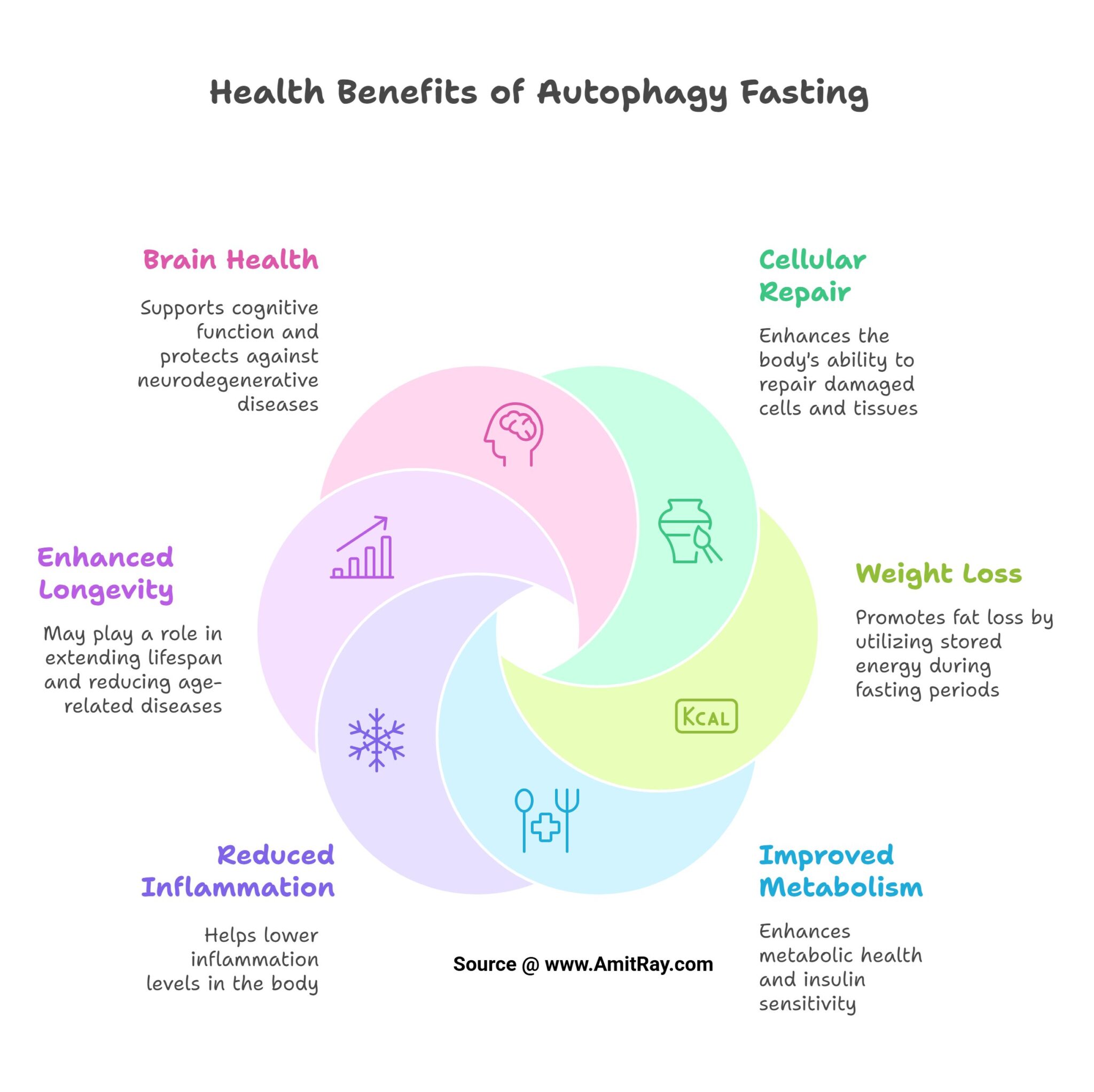
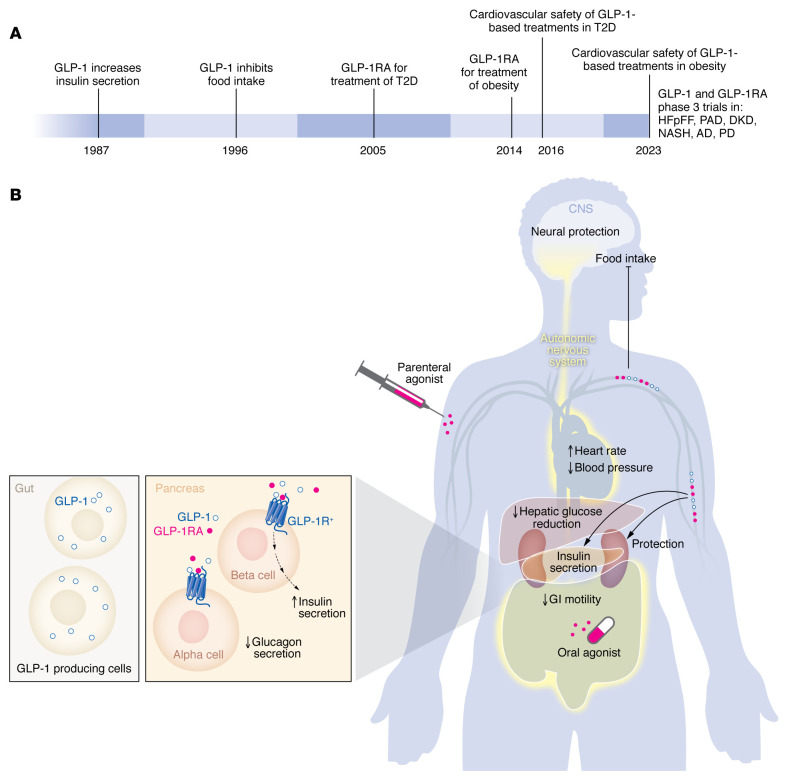
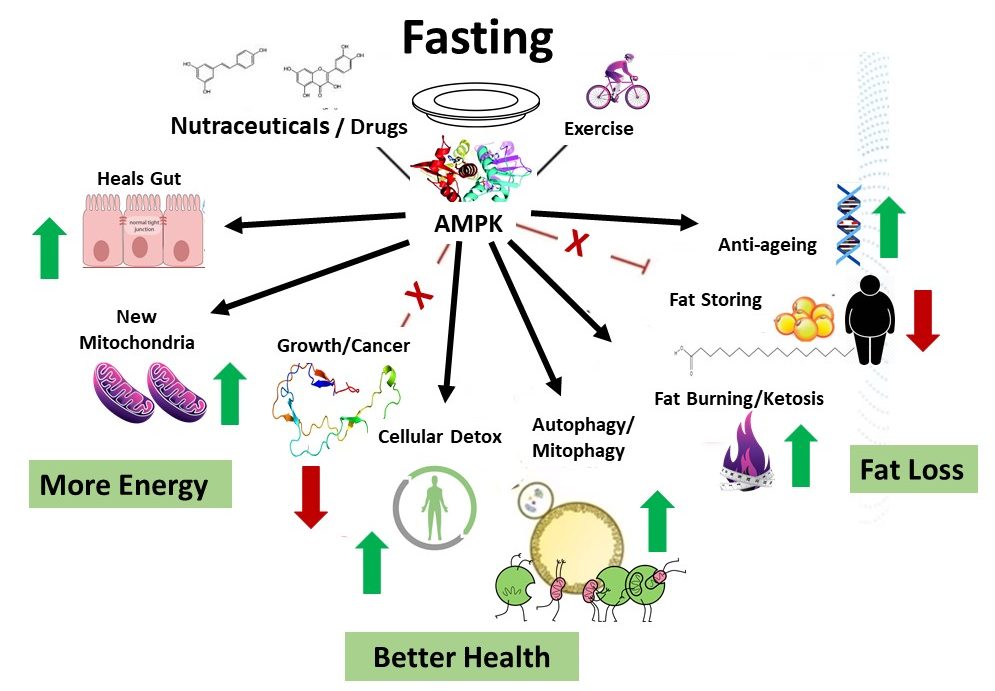
The Impact of GLP-1 on Autophagy During Fasting
Introduction
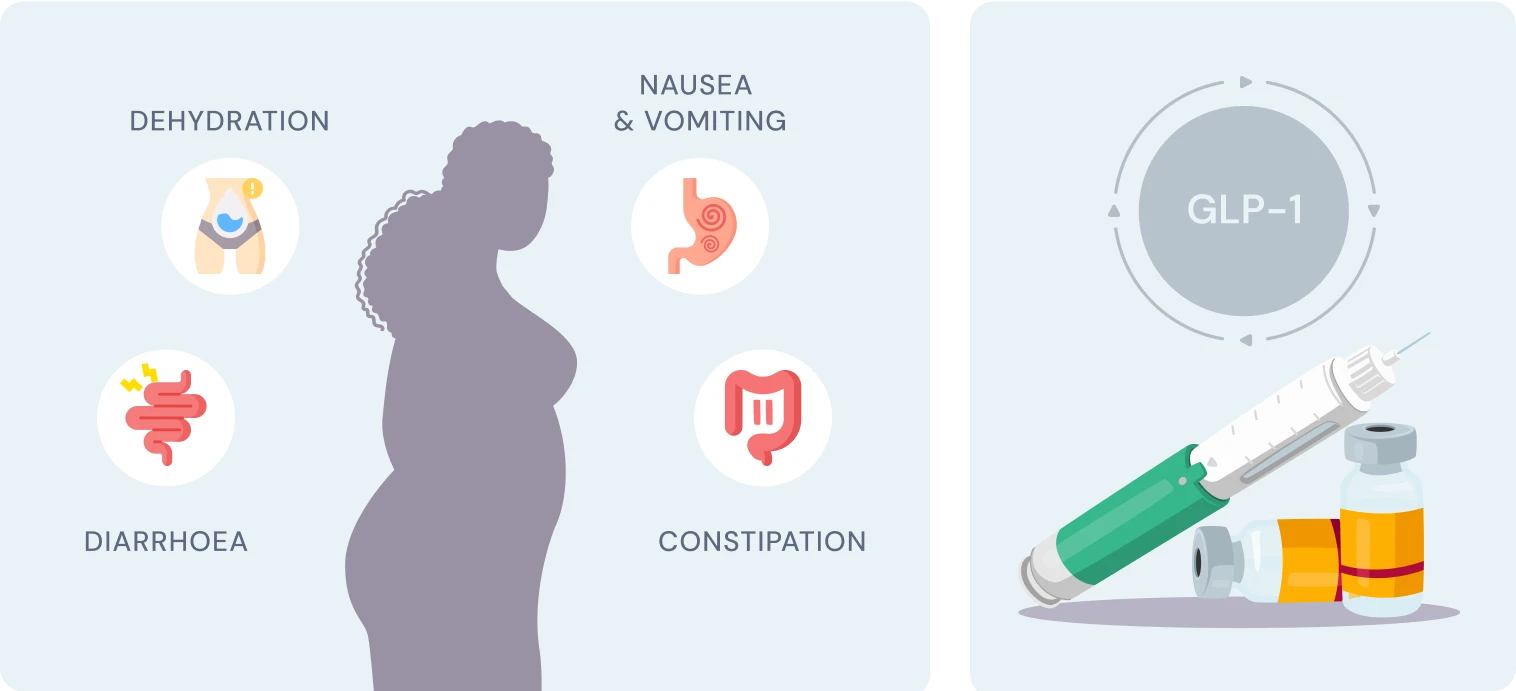
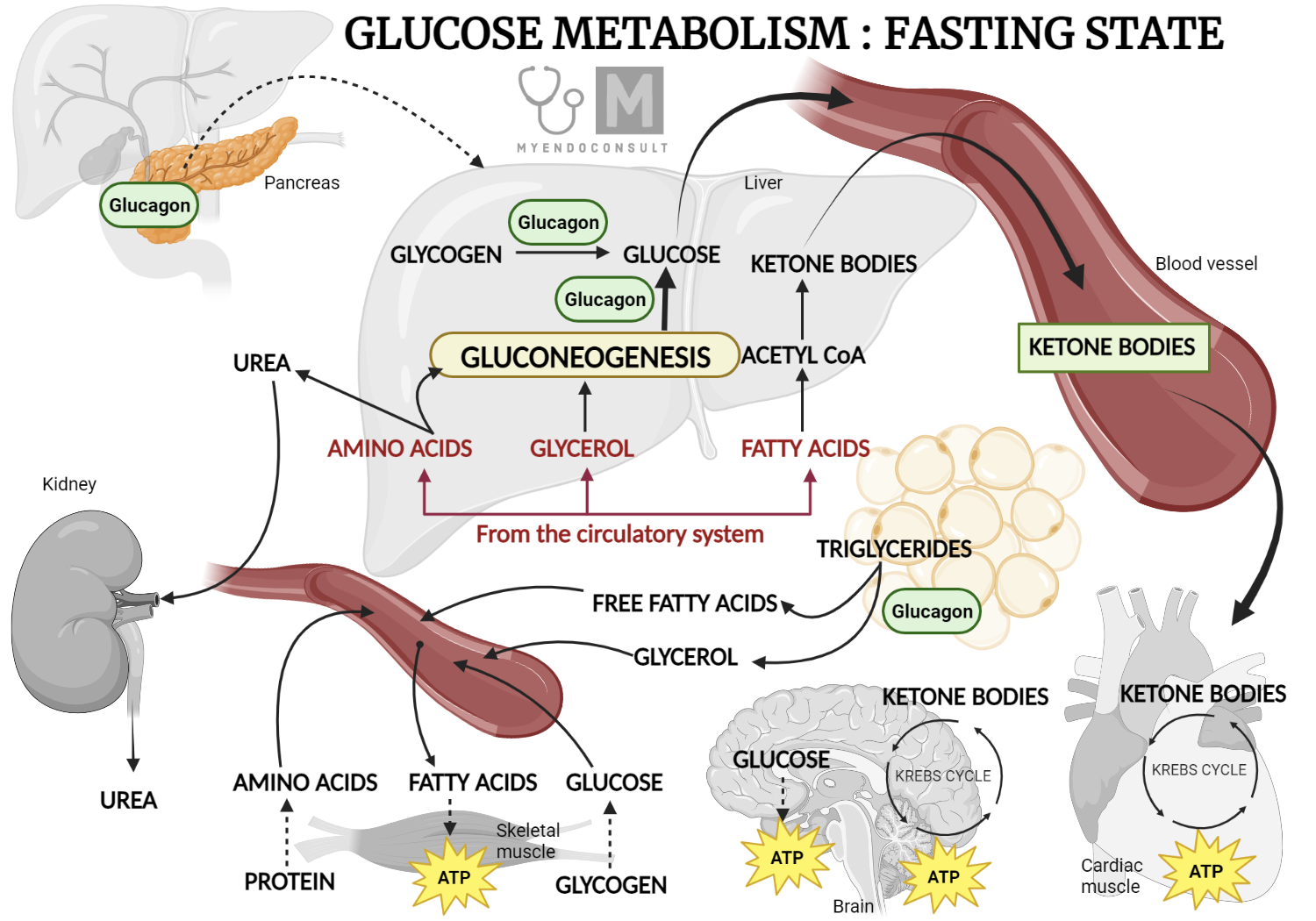
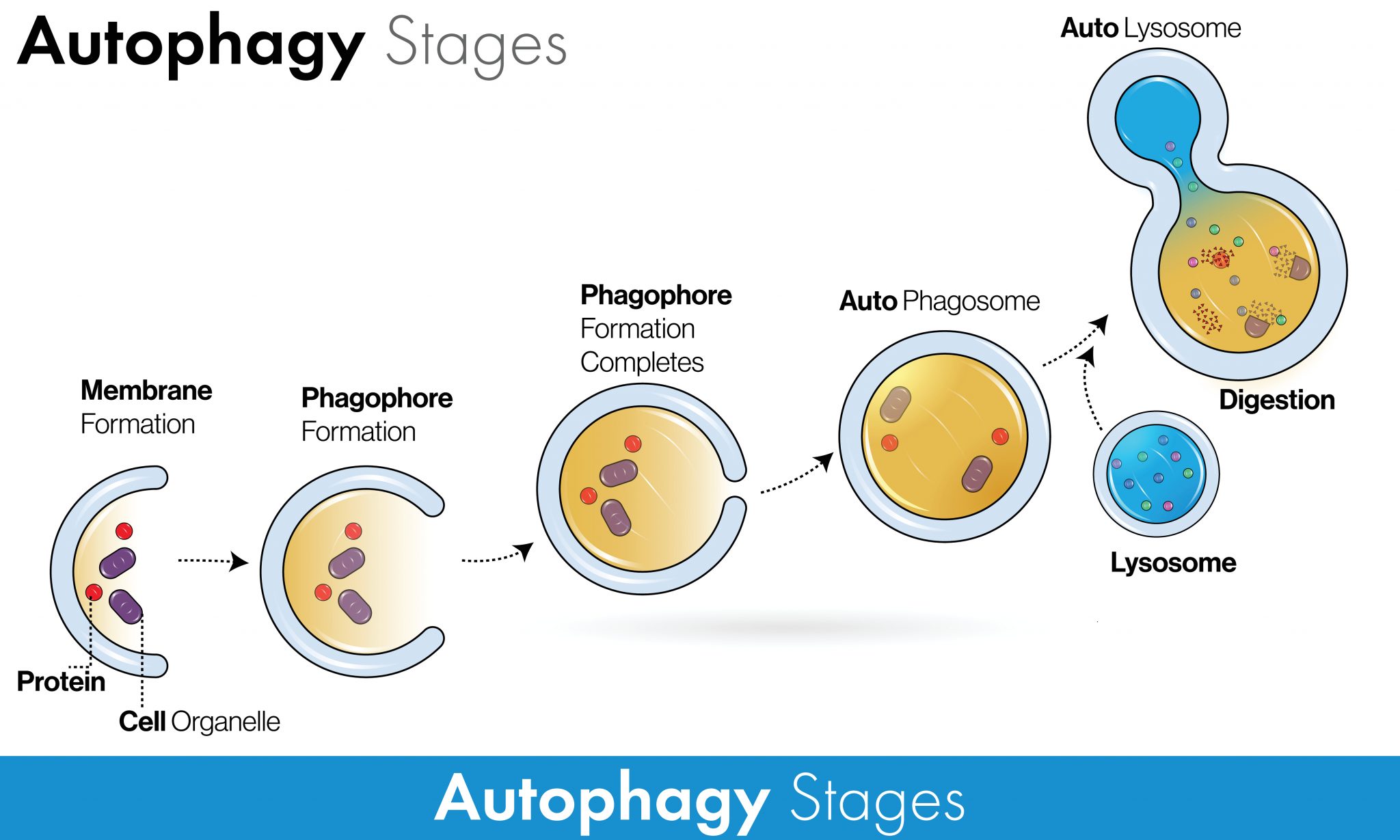
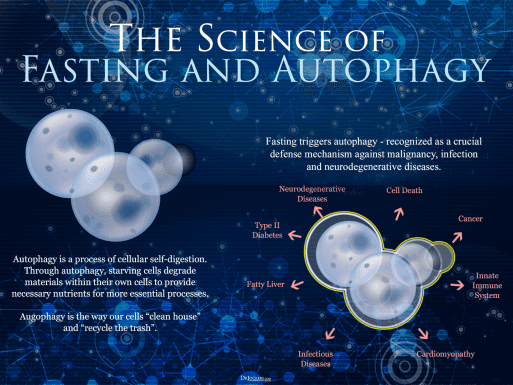
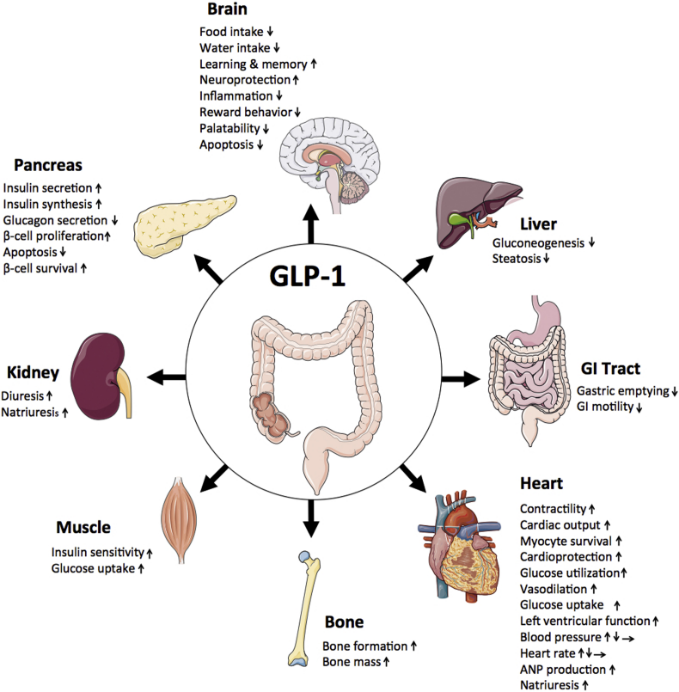
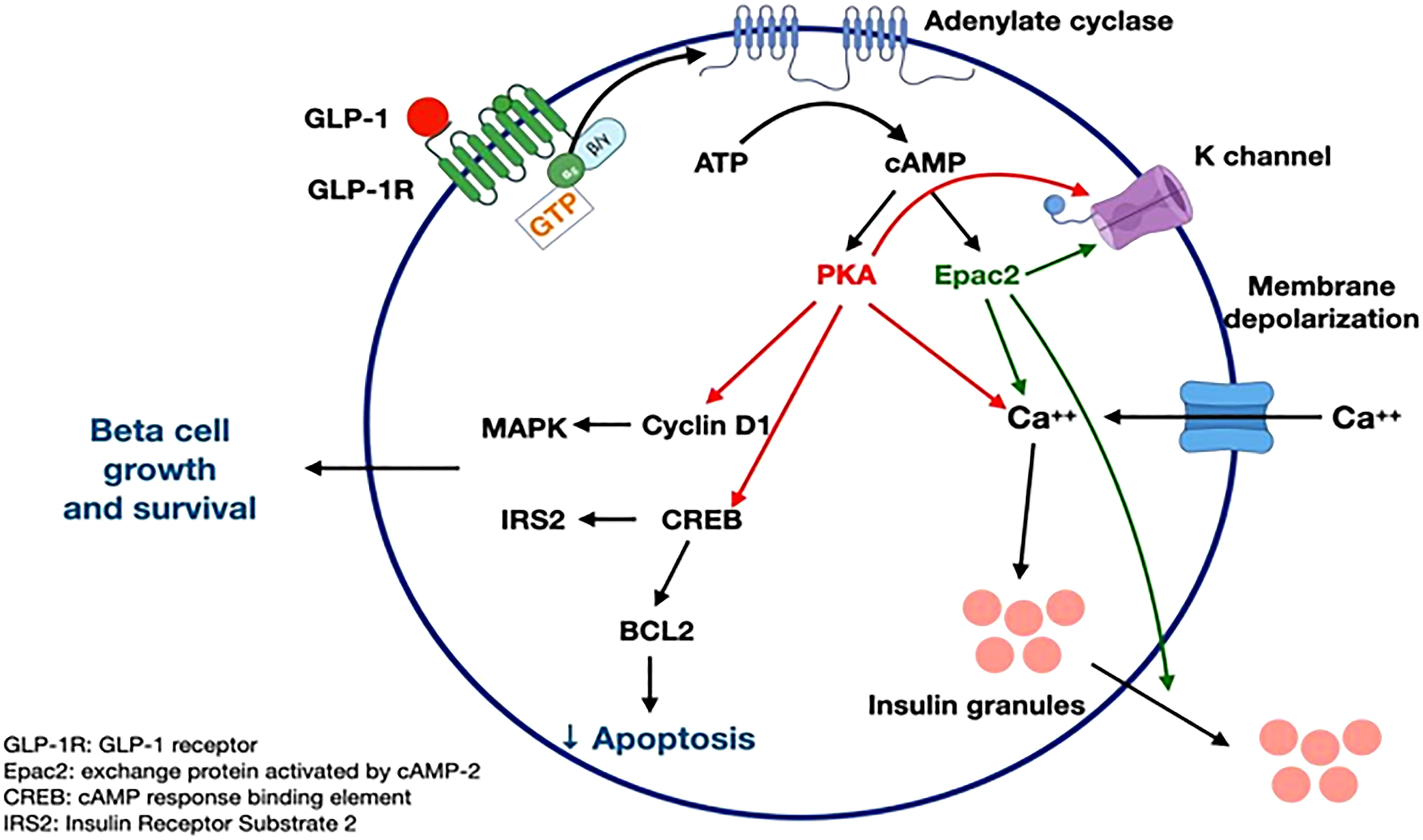
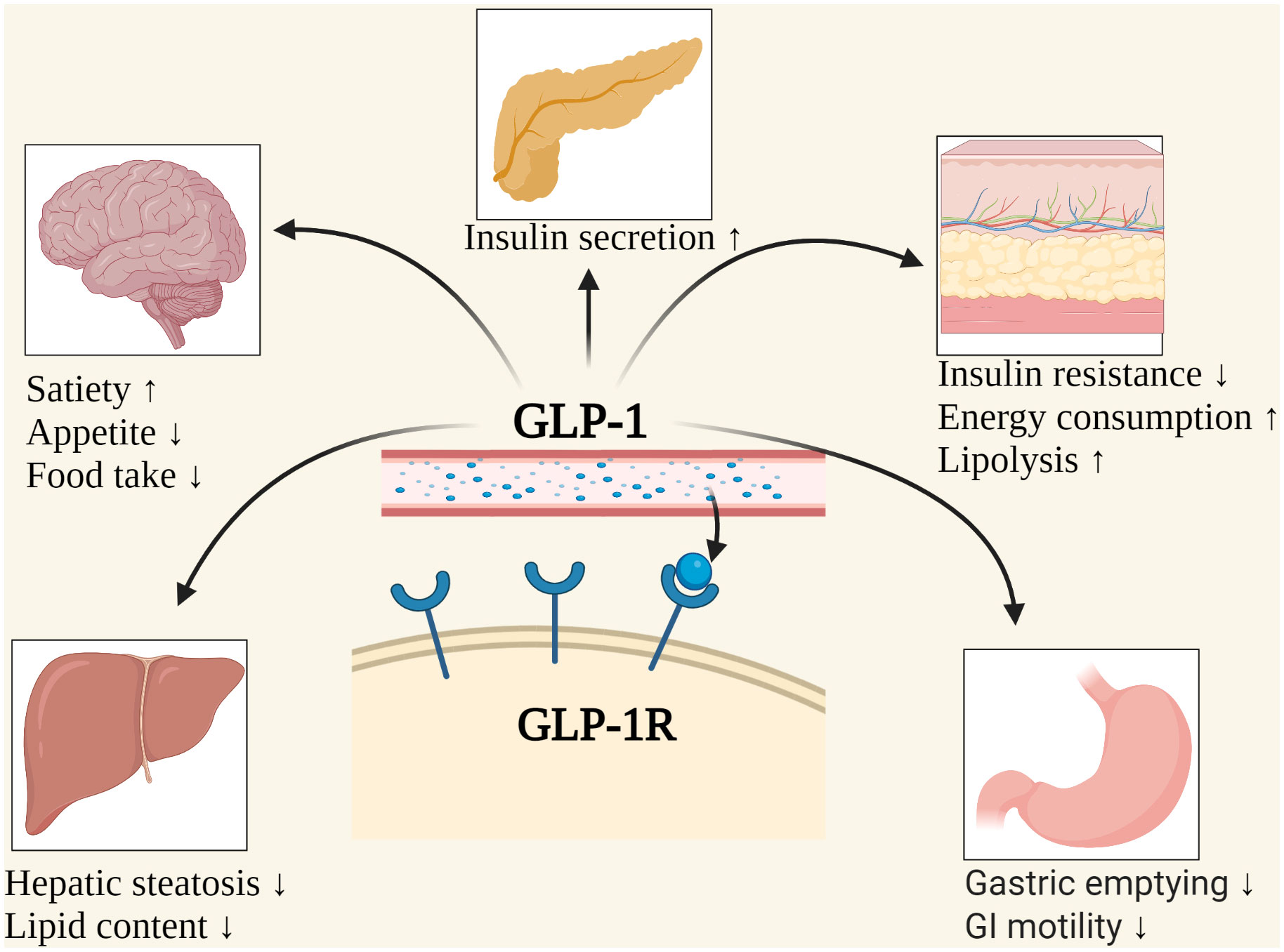
Glucagon-like peptide-1 (GLP-1), a gastrointestinal peptide and central mediator of glucose metabolism, plays a crucial role in regulating blood sugar levels and insulin secretion. When we consume food, GLP-1 is secreted by L cells in the intestine, triggered by nutrient sensing via transporters and G-protein-coupled receptors (GPCRs) (Kim et al., 2022). However, adults with obesity/overweight (OW) or type 2 diabetes mellitus (T2DM) often have altered GLP-1 secretion patterns, which can impact glucose homeostasis (Liebenguth et al., 2018). Autophagy, the body's internal cleanup system, is a vital process that allows cells to recycle damaged or dysfunctional components. During fasting, autophagy is induced, enabling cells to adapt to nutrient deprivation and promoting cellular renewal (Kunos et al., 2017). Emerging evidence suggests that GLP-1 may exert beneficial effects on glucose homeostasis via autophagy-dependent pathways, particularly in pancreatic β-cells and other cell types (Kim et al., 2022).Impact of Fasting on GLP-1 Secretion and Autophagy

Research Findings: The Impact of GLP-1 on Autophagy During Fasting
Research studies have shown that: * GLP-1RAs can enhance insulin sensitivity and reduce body weight, suggesting a potential role in the management of T2DM (Lindsay et al., 2018). * Intermittent fasting can increase autophagy and improve insulin sensitivity, particularly in muscle and adipose tissue (Pedersen et al., 2019). * GLP-1 can modulate autophagy in pancreatic β-cells, improving glucose-stimulated insulin secretion and β-cell function (Kim et al., 2022). * The combination of GLP-1RAs and fasting may upregulate autophagy and improve cellular clearance, leading to enhanced metabolic health (Castillo et al., 2018).Conclusion